A Breakthrough Stem-Cell Approach: Type 1 Diabetes Treatment
Table of contents
AN INTRODUCTION TO MY TRAUMATIC GLACIER OF DIABETES: AN ANALYSIS OF FACTS AND FIGURES
REGENERATION STRATEGIES: UTILIZING TO RESTORE NORMAL HEALTH
Stem Cell Therapy and Cell Transplantation
Trans-Differentiation (Reprogramming)
Getting Current Beta Cells to Multiply
CHALLENGES & THEIR FORESEEABLE FUTURE
Introductions
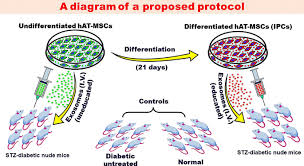
Diabetes, like a second citizen-pain, has morphed itself into this v
The diabetes simply turned from one form of discomfort to another—in this case, low blood sugar, which may slip largely unnoticed into the entire body. Dwindling beta cell activity is the main responsible agent in the disease: they are the cells that produce insulin and are functionally relevant in saving the lives of diabetic patients. Therefore, the aim of future research on diabetes lies indisputably in developing a cure to replace these lost/damaged cells.
🌏 AN INTRODUCTION TO MY TRAUMATIC GLACIER OF DIABETES: AN ANALYSIS OF FACTS AND FIGURES


It is a cruel fact that today’s fastest-escalating diabetes epidemic cries loudly for treatment, not for treatment justification/symptom management.
Prevalence: IDF reports that globally, nearly 590 million adult humans have died of this disease, which, possibly, would, in 2050, see 853 million adult humans entering these numbers with the disease.
Deaths: The disease leads to 4.2 million deaths throughout the world each year.
Type 2 diabetes (T2D) is the dominant case in diabetes, as it is responsible for over 90% of all diabetes cases, while type 1 diabetes (T1D) is only a concern for 5-10% of individuals.
Beta Cell Loss.
In T1D, an autoimmune attack on pancreatic beta cells prevents their proliferation, leading to a gradual decline in insulin production.
The distinction of a sluggish number of beta cells results in a direct loss of function and mass. The onset of hyperglycemia in T2D is more likely due to insulin secretion inhibition.
‘Pancreatic diabetes’ is clinically diagnosed when beta cell loss approaches about 65%.
The present therapy is ultimately only replacement therapy in that it teaches the insulin-appropriate tissues to take over the regimen of replacing cell mass loss or any drug, such as GLP-1 agonists and DPP-4 inhibitors, capable of decreasing sugar levels without treating the real situation itself: functional beta cell mass loss.
💡REGENERATION STRATEGIES: UTILIZING TO RESTORE NORMAL HEALTH
Regeneration of the functional beta cells is the focus of regenerative medicine. Thus, establishing three major linked pathways that point toward this new friend of beta-like cells is the foundation for a future where man could become autonomous.
1. Stem Cell Therapy and Cell Transplantation
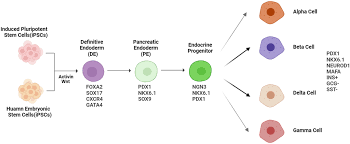
One method of cell therapy using stem cells is to culture many insulin-producing cells for patients’ treatment. Three major connected pathways of prismatic pointing are establishing this future in which man could become self-sufficient: One type of cellular therapy using stem cells will be the in vitro generation of numerous insulin-producing cells intended for future patient-specific transplantation.
Pluripotent stem cells: The stem cells—embryonic hESC or iPS cells cultured from pancreatic tissue—do not exist genetically. They should hopefully be perimenopausal when implanted.
Currently, there are ongoing clinical trials for egregious stem cell-derived islet progenitor cells. The clinical trial recruitment process for ViaCyte and Vertex Pharmaceuticals’ company-based research initiative has started (NCT02239354, NCT03163511). The Phase 1/2 clinical trial of VC-02TM by ViaCyte resulted in insulin-secreting beta-like cells that matured from T1D patients with C-peptide levels exceeding one-third of the study participants.
Islet transplantation demonstrates potential for managing blood sugar levels, although its application remains limited because of insufficient cadaveric islet donations, and patients must stay on immunosuppressive drugs throughout their lives after transplantation
2. Trans-Differentiation (Reprogramming)
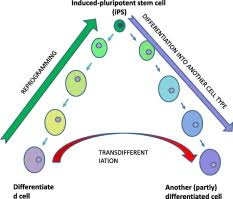
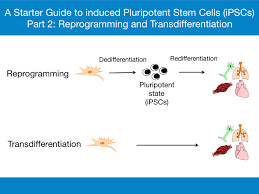
The development of insulin-secreting beta cells from other less important cell types within the pancreas is borrowed from different organs.
Alpha-to-Beta Cell Conversion: These pancreatic alpha cells, which secrete glucagon, were obtained via a lineage including the beta cell. Experimental manipulations can drive alpha lineage cells toward differentiation into functional beta-like cells by using specific transcriptional regulatory factors, mainly by inhibiting Arx.
Ductal Progenitor Cells: Progenitor cells resemble those in growing babies. That’s why it seems logical to test EZH2 inhibitors (GSK126 and Tazemetostat are a couple, and they’re both cancer drugs that the FDA has given the go-ahead to use) to convert duct cells from people with diabetes into cells close to beta cells. Ideally, these revamped cells would then start pumping out insulin when there’s glucose present.
3. Getting Current Beta Cells to Multiply
double-generating, stimulating the remaining beta cells to double-generate additional functional mass.
Small Molecules and Growth Factors: In all, the studies aim to understand molecules that could be good for signalling pathways through which to link intermediacy with them.
A recent approach to inhibiting the DYRK1A kinase has been to increase beta-cell proliferation using novel small molecules, for example, harmine.
These compounds have also acted as incretins studied primarily in diabetes. To this end, the effects of GLP-1 analogues (exenatide and liraglutide) on beta-cell growth and survival have been conclusively demonstrated.
🚧 CHALLENGES & THEIR FORESEEABLE FUTURE
The problems, as regards beta cell regeneration, are several before it is available in an applied form for a cure:
Immune Attack (T1D): If regenerated beta cells are to live and proliferate in T1D socioimmunologically challenging environments, the adapted beta cells should be protected from autoimmune attack. Some possible solutions could presumably involve:
Encapsulation Systems Islets were once encapsulated using the ViaCyte (mostly oozy) device to prevent loss due to hypoxia.
Evasive Cells: It is possible to clothe the transplanted cells so they are ignored by the immune system or, better still, to implant them as immune-tolerant cells.
Immune Reset: Islet transplantation would involve transplanting blood stem cells to create a hybrid immune system to adapt and not recognise new beta cells. This has been a success within a mystifying world of mice.
Functionality and Scalability: In any case, innate stem cell approaches are still developing fields to secure an adequate supply of mature, glucose-responsive cells.
Safety: What has convinced me is that it stops cell proliferation in response to proliferative drugs without overgrowth or showing cross-effects on various cell types.
Or, in some way, antisociety, along with stem cell biology, immune engineering, and the emergence of powerful regenerative molecules, is quickly altering the traditional diabetes therapy concept from a dreamlike, near-fictional, bringing-it-into-reality paradigm to one inching toward a cure.
.

Leave a Reply